Home / JOYSBIO COVID-19 Antigen Rapid Test Kit
* Fields required

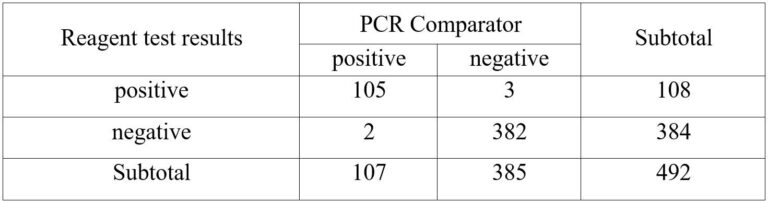
JOYSBIO’s coronavirus Ag test kit was independently evaluated at Centro Diagnostico Delta S.r.l. in Italy between October 2020 and January 2021. A total of 107 positive specimens were tested with JOYSBIO’s COVID-19 Antigen Rapid Test Kit. These specimens were collected from patients who are suspected of COVID-19 with nasal swabs. The coronavirus antigen test kit’s sensitivity and specificity are compared against a CE-IVD marked RT-PCR test kit. This clinical evaluation is conducted under the assumption that SARS-CoV is no longer spreading in the community.
According to the clinical analysis of 492 samples, the detection sensitivity is 98.13%, and the specificity is 99.22%.
The limit of detection (LOD) of this product is 1.6 x 102 TCID50/mL, calculated through a gradient dilution method.
The coronavirus antigen rapid test kit is a lateral flow assay that qualitatively detects the presence of nucleocapsid (N) protein in upper respiratory samples (nasal swabs). This lateral flow assay is designed with the sandwich immunoassay format. When the specimen is added onto the sample pad of a test cassette, coronavirus N protein binds with colloidal gold-labeled SARS-CoV-2 N protein antibody to form an antibody-antigen (Ab-Ag) complex. The Ab-Ag complex is captured by SARS-CoV-2 N protein antibody (Rabbit monoclonal antibody) when migrating to the test line under capillary action. A red-colored band will appear on the test line, which indicates the specimen is COVID-19 nucleocapsid protein positive. No color band will appear on the test line if the specimen does not contain any coronavirus antigen (N protein), or the antigen level is below detection limit.
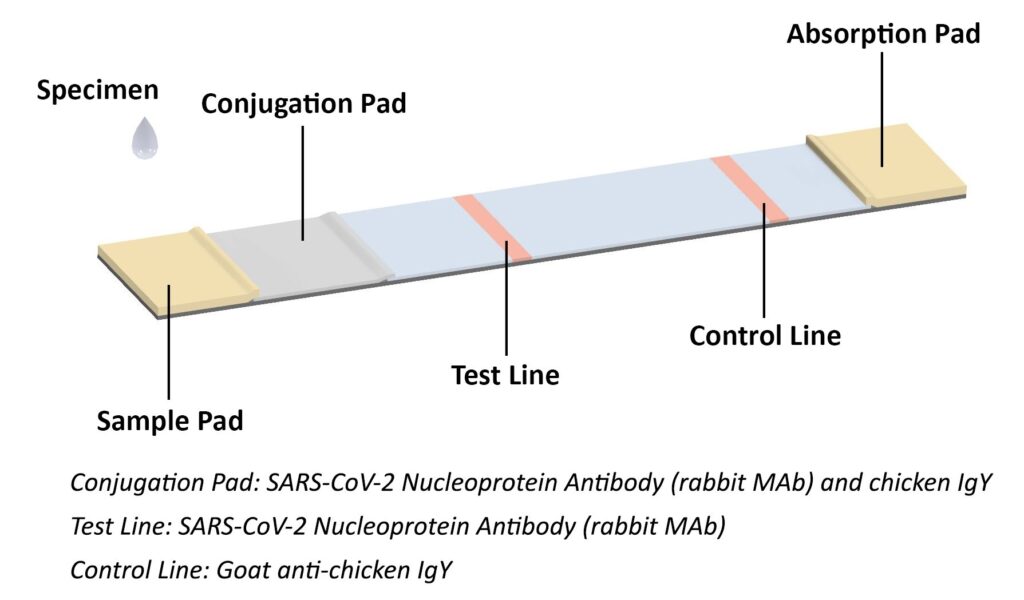
The conjugation pad also contains colloidal gold-labeled chicken IgY, which is captured by Goat anti-chicken IgY on the control line as procedural control. A colored band on the control line represents the proper liquid flow through the cassette; the absence of a colored band on the control line indicates insufficient sample or buffer volume.
Please check Instructions for Use for complete procedure


